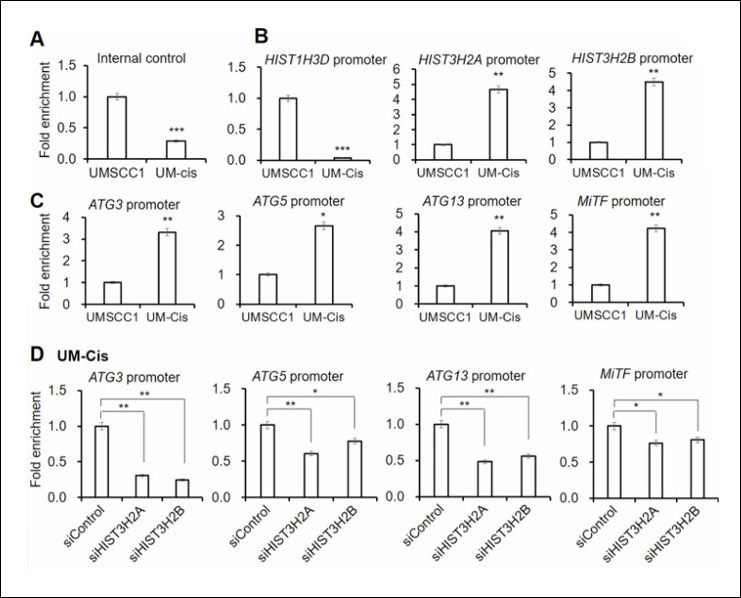
Nimal S et. al. (April 2024). Apigenin and its combination with Vorinostat induces apoptotic-mediated cell death in TNBC by modulating the epigenetic and apoptotic regulators and related miRNAs Sci Rep. 14(1):9540.
This study investigates the anti-TNBC potential of the flavonoid apigenin and its combination with Vorinostat on MDA-MB-231 cells. Apigenin induces apoptotic cell death, inhibits cell migration, and alters cell cycle progression. It reduces class-I HDAC expression, increases HAT activity, and modulates miRNA expression, including upregulating miR-200b and downregulating oncomiR-21. The combination of apigenin and Vorinostat synergistically reduces TNBC cell growth by targeting epigenetic and apoptotic regulators, suggesting a potential treatment strategy for metastatic TNBC.
Products Used: EpiQuik DNA Methyltransferase (DNMT) Activity/Inhibition Assay Kit, EpiQuik Nuclear Extraction Kit, Epigenase HDAC Activity/Inhibition Direct Assay Kit (Colorimetric)
Enjeti AK et. al. (April 2024). Combining 5-azacitidine with the E-selectin antagonist uproleselan is an effective strategy to augment responses in myelodysplasia and acute myeloid leukaemia Br J Haematol.
This study explores the efficacy of combining 5-azacitidine with the E-selectin antagonist uproleselan in treating myelodysplasia and acute myeloid leukaemia (AML). The interaction between AML blasts and the bone marrow microenvironment, mediated by E-selectin, contributes to chemoresistance. Treatment with 5-azacitidine leads to further hypomethylation of genes regulating E-selectin ligands, enhancing E-selectin binding. However, combining 5-azacitidine with uproleselan reverses E-selectin adhesion, prolonging survival in AML mouse models. Clinical evidence supports this combination therapy's potential in higher-risk MDS and AML patients, especially those who are non-responsive to 5-azacitidine alone.
Products Used: MethylFlash Global DNA Methylation (5-mC) ELISA Easy Kit (Colorimetric)
Chen C et. al. (April 2024). Landscape of transcriptome-wide m6A modification in diabetic liver reveals rewiring of PI3K-Akt signaling after physical exercise Chen C et. al. (April 2024).
This article explores how physical exercise affects N6-methyladenosine (m6A) modifications in the liver of diabetic mice. Physical exercise improved liver metabolism and changed RNA m6A levels. It was found that genes related to the PI3K-Akt signaling pathway, crucial for diabetes, were affected. This research highlights how exercise-induced changes in m6A modifications may impact insulin signaling and diabetes progression.
Products Used: EpiQuik m6A RNA Methylation Quantification Kit (Colorimetric)
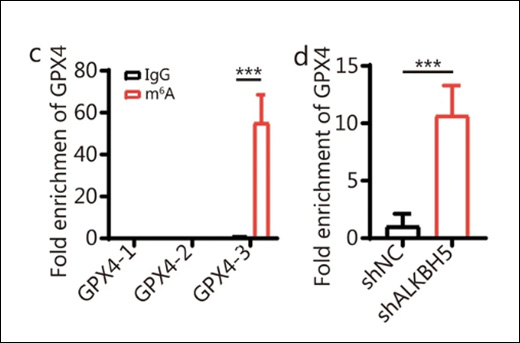
Sun J et. al. (April 2024). Positive GLI1/INHBA feedback loop drives tumor progression in gastric cancer Sun J et. al. (April 2024).
The study investigates the role of GLI1, a key transcription factor in the Hedgehog signaling pathway, in gastric cancer (GC) progression. GLI1 is found to drive GC cell proliferation and metastasis by upregulating INHBA, which in turn activates Smads signaling and enhances GLI1 expression. Disrupting the GLI1-INHBA interaction inhibits GC tumor growth in vivo. Additionally, the study uncovers a novel mechanism involving Helicobacter pylori-induced N6-methyladenosine (m6A) modification in the activation of the GLI1 pathway, suggesting potential therapeutic targets for GC treatment.
Products Used: EpiQuik m6A RNA Methylation Quantification Kit (Colorimetric)
Xie B et. al. (April 2024). ZC3H13 promotes ITGA6 m6A modification for chronic obstructive pulmonary disease progression Cell Signal. :111190.
The study examines the role of the m6A methylation transferase ZC3H13 in chronic obstructive pulmonary disease (COPD) progression. ZC3H13 is upregulated in COPD and correlates with the expression of ITGA6, an integrin involved in inflammation and fibrosis. Knockdown of ZC3H13 improves COPD symptoms in cellular and mouse models by reducing inflammation, apoptosis, and epithelial-mesenchymal transition (EMT). Mechanistically, ZC3H13 enhances ITGA6 expression and mRNA stability through m6A modification, promoting bronchial epithelial cell inflammation and fibrosis. Targeting the ZC3H13/ITGA6 axis could be a potential therapeutic approach for COPD.
Products Used: EpiQuik m6A RNA Methylation Quantification Kit (Colorimetric)
Yang Y et. al. (April 2024). Hirschsprung's disease: m6A methylase VIRMA suppresses cell migration and proliferation by regulating GSK3β Yang Y et. al. (April 2024).
The study investigates the role of the m6A methylase VIRMA in Hirschsprung's disease (HSCR). VIRMA is downregulated in narrow-segment colon tissue of HSCR patients, and its silencing inhibits cell proliferation and migration. Mechanistically, VIRMA upregulates the expression of GSK3β by inhibiting its mRNA degradation, and GSK3β mediates VIRMA-driven cell migration and proliferation. These findings suggest that VIRMA suppresses cell migration and proliferation in HSCR by regulating GSK3β expression, highlighting VIRMA as a potential therapeutic target for HSCR.
Products Used: EpiQuik m6A RNA Methylation Quantification Kit (Colorimetric)
Li Z et. al. (April 2024). Histone H3K27 Demethylase SlJMJ3 Modulates Fruit Ripening in Tomato Plant Physiol.
The article investigates the role of the histone lysine demethylase SlJMJ3 in tomato fruit ripening. SlJMJ3, a member of the KDM4/JHDM3 demethylase family, accelerates fruit ripening when overexpressed and delays it when knocked down. SlJMJ3 regulates ripening-related genes involved in ethylene biosynthesis and response, carotenoid metabolism, cell wall modification, transcriptional control, and DNA methylation modification. SlJMJ3 directly binds to the promoters of these genes, reducing H3K27me3 levels and upregulating their expression. These findings suggest that SlJMJ3 plays a crucial role in tomato fruit ripening by acting as an epigenetic regulator.
Products Used: EpiQuik Total Histone Extraction Kit
Wang Z et. al. (April 2024). TMEM9 promotes lung adenocarcinoma progression via activating the MEK/ERK/STAT3 pathway to induce VEGF expression Cell Death Dis. 15(4):295.
The study investigates the role of Transmembrane protein 9 (TMEM9) in lung adenocarcinoma (LUAD) progression. TMEM9 is significantly upregulated in LUAD tissues and correlates with poor prognosis. Downregulation of TMEM9 suppresses tumor growth and metastasis, as well as reduces angiogenesis in vitro and in vivo. TMEM9 upregulates VEGF expression, and blocking VEGF reverses the angiogenic and migratory effects of TMEM9. Mechanistically, TMEM9 activates the MEK/ERK/STAT3 pathway to induce VEGF expression. These findings suggest that targeting the TMEM9/MEK/ERK/STAT3/VEGF pathway could be a potential therapeutic strategy for LUAD.
Products Used: TMEM9 Polyclonal Antibody



 Cart (0)
Cart (0)